#Schrodinger wave equation plus
In words, this equation could be described as “total energy equals kinetic energy plus potential energy”. Is the Laplacian operator, and ψ is the wave function (more precisely in this context, it is called the “position–space wave function”). Where m is the particle's mass, V is its potential energy, However, the complex amplitudes taken by the two wave functions which represent the electron passing each slit do follow a law of precisely the form expected Ψ (total) = ψ (first) + ψ (second), and the calculations agree with experiment. Thus, when not watching the electron, the particle cannot be said to go through either slit and this simplistic explanation does not work. However, it is impossible to observe which slit is passed through without altering the electron. In the classic double–slit experiment where electrons are fired randomly at two slits, an intuitive interpretation is that P (hit either slit) = P (hit first slit) + P (hit second slit), where P (event) is the probability of that event. Although ψ is a complex number, | ψ | 2 is real, and corresponds to the probability density of finding a particle in a given place at a given time, if the particle's position is measured. The most common symbol for a wave function is ψ (psi).
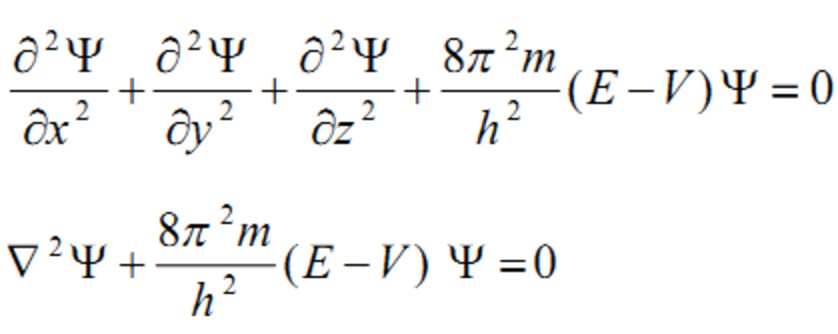
The wave function behaves qualitatively like other waves, like water waves or waves on a string. The laws of quantum mechanics (the Schrodinger equation) describe how the wave function evolves over time. The values of a wave function are complex numbers and, for a single particle, it is a function of space and time. The values taken by a normalized wave function Ψ at each point x are probability amplitudes, since | Ψ(x) | 2 gives the probability density at position x. Probability amplitude is a complex number whose modulus squared represents a probability or probability density. Wave function is a probability amplitude in quantum mechanics describing the quantum state of a particle and how it behaves.
